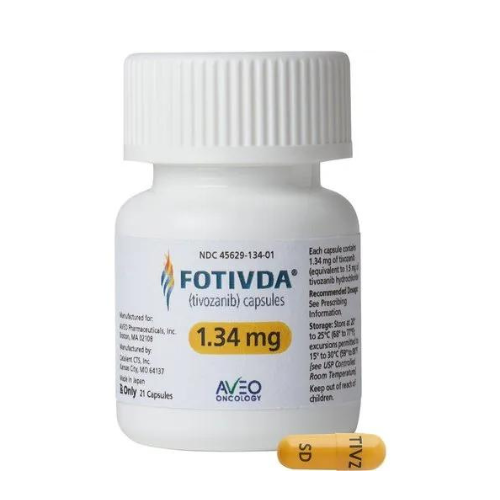
About Tivozanib
- Fotivda (Tivozanib) contains the active ingredient tivozanib and received FDA approval on July 6, 2023, for treating adults with advanced renal cell carcinoma (RCC) who have relapsed or refractory disease.
- Tivozanib, is an oral VEGF receptor tyrosine kinase inhibitor. It works by inhibiting blood vessel growth that supplies nutrients and oxygen to the tumor, thereby slowing or halting cancer cell growth.
- This mechanism suggests promising results in managing relapsed or refractory advanced RCC, indicating a favorable success rate in treating this type of kidney cancer.
Strength:
- Fotivda capsules of 1.34 mg and 0.89 mg
Recommended Dosage:
Fotivda capsules are designed to be taken orally and can be consumed with or without food. However, it is advisable to consult your healthcare provider for more detailed information regarding dosage instructions tailored to your specific medical condition.
They can provide personalized guidance based on factors such as your overall health, medical history, and any other medications you may be taking. Always follow your doctor’s recommendations carefully to ensure the safe and effective use of the drug.
- The recommended dose of 1.34 mg of Fotivda is to be taken once daily, with or without food, for 21 days followed by 7 days of treatment, constituting a 28-day cycle, until disease progression or unacceptable toxicity.
- Dose interruptions and/or reductions may be necessary to manage adverse reactions effectively. In patients with moderate hepatic impairment, the dose should be reduced to 0.89 mg for 21 days on treatment followed by 7 days off treatment.
Important:
Before starting the treatment, it’s crucial to discuss any existing medications, medical conditions, pregnancy status, or plans for breastfeeding with your healthcare provider. This information enables your doctor to assess potential drug interactions and tailor the treatment plan to your individual needs, prioritizing your safety and well-being. For further clarification and guidance, consult your doctor for personalized information and advice.
Warnings & Precautions
- Avoid using strong CYP3A inducers concomitantly.
- Before initiating Fotivda treatment, it’s crucial to control blood pressure. Monitor patients for hypertension and manage it as necessary. If hypertension persists despite anti-hypertensive medications, consider reducing the Fotivda dose.
- Throughout treatment, closely monitor for signs or symptoms of cardiac failure, cardiac ischemia, arterial thromboembolic events, and venous thromboembolic events, especially in patients at increased risk.
- Permanently discontinue the medicine for severe arterial or venous thromboembolic events.
- Monitor patients at risk of or with a history of bleeding for hemorrhagic events. Proteinuria should be monitored throughout treatment, and for moderate to severe cases, consider dose reduction or temporary interruption.
- Monitor thyroid function before and during the treatment. Withhold Fotivda before elective surgery for at least 24 days and 2 weeks following major surgery and adequate wound healing.
- Discontinue the drug if signs or symptoms of reversible posterior leukoencephalopathy syndrome occur.
- Advise patients of potential embryo-fetal toxicity and the need for effective contraception.
- Additionally, caution patients allergic to tartrazine, present in the 0.89 mg Fotivda capsule, of possible allergic reactions.
Common Fotivda Side Effects:
- Fatigue
- Hypertension
- Diarrhea
- Decreased appetite
- Nausea
- Dysphonia
- Hypothyroidism
- Cough
- Stomatitis
Laboratory abnormalities of Grade 3 or 4, occurring in ≥5% of cases, included:
- Decreased sodium
- Increased lipase
- Decreased phosphate
Use in Specific Population
- Fotivda has the potential to cause fetal harm in pregnant women based on animal studies and its mechanism of action.
- There is insufficient data on the presence of tivozanib in human milk, its effects on breastfed infants, or milk production. Therefore, lactating women are advised not to breastfeed during the treatment and for one month after the last dose.
- Prior to initiating treatment, verify the pregnancy status of females of reproductive potential and advise them to use effective contraception throughout treatment and for one month after discontinuation.
- Male patients with female partners of reproductive potential should also use effective contraception during and for one month after the treatment. Animal studies suggest that this medicine may impair fertility in both females and males of reproductive potential.
- The safety and efficacy of Fotivda in pediatric patients have not been established. No dosage adjustment is necessary for patients with mild to severe renal impairment, but the recommended dosage for those with end-stage renal disease has not been determined.
- Similarly, the recommended dosage for patients with severe hepatic impairment has not been established. Excessive intake of Fotivda can lead to severe hypertension and hypertensive crisis, which may be fatal.
Storage and Handling
- Store Fotivda at 20ºC to 25ºC (68ºF to 77ºF), with excursions permitted from 15ºC to 30ºC (59ºF to 86ºF).
- Keep the medicine protected from moisture.
- Do not freeze the medicine.
- Keep the medicine away from pets and children.
Our pharmaceutical procurement process is carefully organized, comprising four essential stages to guarantee a smooth and efficient experience:
- Inquiry: Our dedicated Named Patient Access Program ensures swift responses to medication inquiries, typically within 24 hours, offering comprehensive assistance.
- Verification: Sansfro ensures the availability and authorization of medications, particularly for patients seeking drugs not easily accessible in their home countries. We meticulously validate prescriptions and medical information with precision, strictly adhering to compliance standards.
- Procurement: Following thorough verification, our team utilizes our extensive supplier network to obtain the necessary medication. We negotiate for competitive quotes and manage the efficient processing of orders to ensure seamless procurement.
- Secure Dispatch: After finalizing the quote, we seamlessly coordinate the secure dispatch of your medication. Our logistics specialists are on hand to track consignments. Upholding the integrity of medication provision, we strictly follow Standard Operating Procedures within the Named Patient Import Program.
For smooth medication importation, patients are required to submit the following documentation:
- A valid prescription copy.
- Identification records.
- Details of the primary healthcare provider.
- Current residential address.
Upon receipt of all necessary documents, the Sansfro team promptly begins the application process for the import license. This license is essential for acquiring the required medication, pending government approval.
No news found.
About Sansfro
Sansfro is committed to bridging the healthcare gap. Our purpose is to bring hope and healing to patients suffering from rare diseases by linking them with life-saving treatments through our Named Patient Program. Join us in our quest for a better and healthier world.
Know More...FAQ'S
What is Tivozanib?
Fotivda (tivozanib) is a prescription medicine used to treat adults with advanced kidney cancer (advanced renal cell carcinoma or RCC) that has been treated with 2 or more prior medicines and has come back or did not respond to treatment.
What are the common side effects of Fotivda?
The most common side effects of Fotivda include tiredness, diarrhea, decreased appetite, nausea, hoarseness, low levels of thyroid hormones, cough, mouth sores, and decreased blood levels of salt (sodium) and phosphate. Other side effects may include vomiting weakness or lack of energy.
Which medical conditions should I disclose to my healthcare provider before starting Fotivda treatment?
Before starting Fotivda treatment, it’s crucial to disclose specific medical conditions to your healthcare provider. These include high blood pressure, heart failure history, previous blood clot occurrences, bleeding disorders, thyroid problems, liver issues, unhealed wounds, planned or recent surgeries, allergies to FD&C Yellow No.5 (tartrazine) or aspirin, and pregnancy or plans to conceive. Adequate disclosure allows your healthcare provider to customize your treatment plan and monitor you effectively throughout the therapy.
What is the connection between the Fotivda capsule and the Tivozanib capsule?
Fotivda and Tivozanib are different names for the same medication. Fotivda is a brand name for the drug Tivozanib, which comes in the form of capsules.
What is the Fotivda price in India?
For the precise price of Fotivda in the Indian market, several factors like import duties, taxes, exchange rates, currency fluctuations, and supply-demand dynamics come into play. To get accurate pricing details that suit your requirements, we suggest reaching out to our Patient Support Team at (+91) 93157 05373 or via email at help@sansfro.com. Our dedicated team of experts is ready to offer personalized assistance and reliable information to address your queries effectively. Don’t hesitate to contact us for any assistance regarding the price of Fotivda in India.
How can I buy Fotivda online?
If you’re considering buying Fotivda online, especially if it’s only available in the US and Europe, we recommend contacting the Sansfro Health team or other reputable companies experienced in importing medications from these regions. This ensures a reliable and secure procurement process. Seeking advice from experienced professionals is crucial, and Sansfro Health is a reputable organization dedicated to facilitating access to authentic pharmaceutical products.

Dr Anchal Aryan
Medical counselor
Patient Stories
Word Wide Delivery:
India, Andorra, Argentina, Australia, Austria, Azerbaijan, Bahrain, Brazil, Bulgaria, Cambodia, Canada, Chile, Colombia, Costa Rica, Croatia, Cyprus, Denmark, Dominican Republic, Estonia, Finland, France, Georgia, Germany, Ghana, Greece, Guatemala, Iraq, Ireland, Israel, Italy, Jamaica, Japan, Jordan, Kenya, Kuwait, Latvia, Lebanon, Libya, Lithuania, Malawi, Mexico, Montenegro, Nepal, Netherlands, New Zealand, Nigeria, Norway, Oman, Pakistan, Paraguay, Peru, Poland, Qatar, Romania, Saudi Arabia, Serbia, Singapore, Slovenia, Spain, Sri Lanka, Sweden, Switzerland, United Arab Emirates, United Kingdom, United States, Venezuela, Zimbabwe, Afghanistan, Albania, Algeria, American Samoa, Angola, Anguilla, Antarctica, etc.