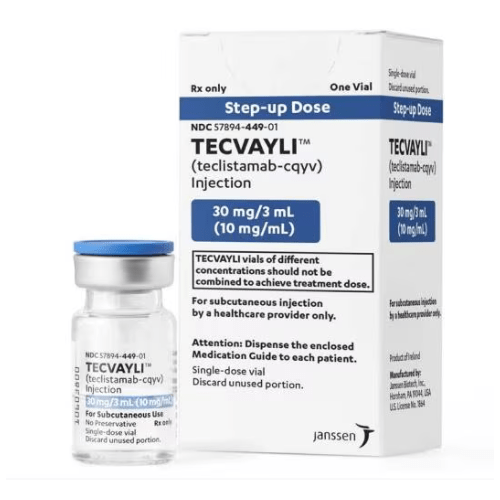
About Tecvayli
- Tecvayli received approval from the US FDA administration on October 25, 2022.
- Tecvayli is an anti-BCMA antibody which belongs to the drug class known as miscellaneous antineoplastics. Tecvayli is a bispecific treatment targeting B-cell maturation antigen (BCMA) and CD3 T-cells.
- This medicine is used in adults with relapsed or refractory multiple myeloma who have undergone a minimum of four prior treatments, including a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38 monoclonal antibody.
- Tecvayli aids the immune system in battling cancer cells by attaching to them, enabling the immune system to identify and combat the cancerous cells.
- Multiple myeloma is a cancer that originates in abnormal plasma cells (type of WBC) in the bone marrow, leading to the overproduction of non-functional antibodies and impacting bone health, immune function, and blood cell production.
- Symptoms such as bone pain, fatigue, frequent infections, anemia, and weakened bones.
- Early detection and appropriate medical intervention are crucial for managing and treating multiple myeloma effectively.
- Tecvayli is available only through a restricted program called the Tecvayli Risk Evaluation and Mitigation Strategy (REMS).
Strength
Injection: 30 mg/3 mL (10 mg/mL); 153 mg/1.7 mL (90 mg/mL) in a single-dose vial
Recommended Dosage
Administering subcutaneous injections is a medical procedure for trained healthcare professionals, involving strict aseptic techniques, continuous monitoring for adverse reactions, and proper disposal of used materials according to medical waste guidelines.
- The recommended dosing schedule for Tecvayli involves a step-up dosing schedule starting on Day 1 with a dose of 0.06 mg/kg, followed by a step-up dose on Day 4 at 0.3 mg/kg. On Day 7, the first treatment dose is administered at 1.5 mg/kg.
- Subsequent treatment doses of 1.5 mg/kg are given once weekly, starting one week after the first treatment dose and continuing weekly thereafter. This dosing regimen aims to optimize the therapeutic effects of Tecvayli.
- Following each dose within the Tecvayli step-up dosing schedule, patients should be hospitalized for 48 hours.
- During this period, it is essential to administer pretreatment medications as recommended to ensure patient safety and manage potential side effects.
- This hospitalization and pretreatment regimen contribute to a comprehensive approach in optimizing the administration of Tecvayli and monitoring patient well-being.
Important
If you are currently taking any medications, please inform your doctor before starting the new medicine. Encourage patients to communicate all concurrent medications, encompassing prescription drugs, over-the-counter medications, vitamins, and herbal products, to their healthcare providers.
Refrain from driving or operating heavy machinery for 48 hours after completing the Tecvayli “step-up dosing schedule” and throughout the entire treatment. If new neurological symptoms develop during Tecvayli treatment, avoid driving or operating machinery until the symptoms resolve.
Warnings & Precautions
- Patients receiving Tecvayli are at risk of experiencing Cytokine Release Syndrome (CRS), which can be life-threatening.
- Initiate treatment with the Tecvayli step-up dosing schedule to reduce the risk of CRS. In the event of CRS, withhold Tecvayli until it resolves or permanently discontinue based on the severity of the reaction.
- Additionally, neurologic toxicity, including Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS), may occur and could lead to serious or life-threatening reactions.
- Patients should be closely monitored for signs or symptoms of neurologic toxicity, including ICANS, during Tecvayli treatment.
- Similar to CRS, withhold Tecvayli until neurologic toxicity resolves or permanently discontinue based on severity. It is crucial to promptly inform your physician if you experience any symptoms related to these conditions.
- ELAHERE may affect the liver, leading to serious issues. So it is recommended to monitor liver enzymes and bilirubin regularly during treatment.
- Life-threatening infections may occur, so monitor for signs, treat appropriately, and withhold ELAHERE in cases of active infection during the step-up dosing schedule.
- Neutropenia is possible; monitor blood counts regularly.
- Systemic and local injection site reactions can occur; consider withholding or permanent discontinuation based on severity.
- Patients should promptly inform their physician of any related symptoms.
Common Tecvayli Side Effects:
- Pyrexia
- Cytokine release syndrome
- Musculoskeletal pain
- Injection site reaction
- Fatigue
- Upper respiratory tract infection
- Nausea
- Headache
- Pneumonia
- Diarrhea
- Decreased lymphocytes
- Decreased neutrophils
- Decreased white blood cells
- Decreased hemoglobin
- Decreased platelets
Use in Specific Population
- The use of Tecvayli may potentially harm a developing fetus, based on animal studies. Hence, it is essential to inform your doctor if you are pregnant, planning pregnancy, or suspect pregnancy before starting treatment.
- Tecvayli is associated with hypogammaglobulinemia, indicating a need to assess immunoglobulin levels in newborns of mothers treated with the drug.
- Breastfeeding while on Tecvayli is not advised due to potential serious adverse reactions in the child; patients should refrain from breastfeeding during treatment and for 5 months after the last dose.
- Females of reproductive age should utilize effective contraception during treatment and for 5 months post-Tecvayli.
- Safety and efficacy remain unestablished in pediatric patients.
- No significant differences in safety or effectiveness were observed in patients aged 65 to 74 compared to younger individuals.
Storage and Handling
- Store Tecvayli at room temperature of 2°C to 8°C (36°F to 46°F).
- Protect Tecvayli from light and moisture.
- Do not freeze.
- Avoid storing Tecvayli in the place where it may be exposed to heat or humidity.
- Keep Tecvayli out of the reach of children and pets.
To make it easier for patients to obtain necessary prescription drugs, Sansfro has developed a comprehensive four-step process:
- Medication Inquiry: If a patient has questions concerning a particular medication, Sansfro’s Named Access Programme Support team will respond to them 24 hours.
- Verification Process: Sansfro verifies the availability and approval of medications while ensuring that prescriptions and medical records are accurate and up to date.
- Medication Procurement: Sansfro’s employees promptly contact their supplier network to secure the medication after it has been verified.
- Safe Delivery: Sansfro makes arrangements for safe medication delivery after the price is agreed upon. Their logistics department makes sure that standard operating procedures are followed for distribution that is both lawful and safe.
Sansfro streamlines the medication import process by initially requesting specific documents from patients:
- An authorized copy of the prescription.
- Proof of the patient’s identification.
- Information about the prescribing physician.
- The patient’s current home address.
Upon receiving these documents, the Sansfro team takes the necessary steps to prepare and submit the application for an import license. The acquisition of this license, contingent on government approval, is a pivotal prerequisite for obtaining the prescribed medication.
No news found.
About Sansfro
Sansfro is committed to bridging the healthcare gap. Our purpose is to bring hope and healing to patients suffering from rare diseases by linking them with life-saving treatments through our Named Patient Program. Join us in our quest for a better and healthier world.
Know More...FAQ'S
What is Tecvayli’s mechanism of action?
Tecvayli activates the immune system by binding to the CD3 receptor on T-cells and the B-cell maturation antigen (BCMA) on multiple myeloma cells and certain healthy B-lineage cells.
What are the side effects associated with Tecvayli?
Mild side effects observed with Tecvayli encompass headache, fever, chills, mild infections like the common cold or urinary tract infection (UTI), muscle and joint pain, back, arm, or leg pain, injection site reactions, fatigue, and diarrhea. It’s essential to be aware of these potential effects and promptly communicate any concerns to your healthcare provider.
Why is breastfeeding not recommended with Tecvayli injection?
Breastfeeding is not recommended during Tecvayli injection treatment and for 5 months after the last dose due to potential adverse effects on the nursing child. It’s important to discuss alternative feeding options with your healthcare provider.
How do Tecvayli and Teclistamab-cqyv differ?
Tecvayli is the branded version of the medication, whereas Teclistamab-cqyv represents the generic name or active pharmaceutical ingredient (API) of the same drug. Essentially, they refer to identical medications with distinctions in nomenclature.
How can I Buy Tecvayli online?
Procuring Tecvayli online has become more convenient. To obtain this medication, which is presently accessible in the US and Europe, consider contacting the SANSFRO team or other organizations specializing in importing medications from these regions.
What is the Tecvayli cost in India?
Tecvayli price in India is contingent on specific product requirements. For detailed information, reach out to our Patient Support Team at (91) 93157 05373 or via email at help@sansfro.com.

Dr Anchal Aryan
Medical counselor
Patient Stories
Word Wide Delivery:
India, Andorra, Argentina, Australia, Austria, Azerbaijan, Bahrain, Brazil, Bulgaria, Cambodia, Canada, Chile, Colombia, Costa Rica, Croatia, Cyprus, Denmark, Dominican Republic, Estonia, Finland, France, Georgia, Germany, Ghana, Greece, Guatemala, Iraq, Ireland, Israel, Italy, Jamaica, Japan, Jordan, Kenya, Kuwait, Latvia, Lebanon, Libya, Lithuania, Malawi, Mexico, Montenegro, Nepal, Netherlands, New Zealand, Nigeria, Norway, Oman, Pakistan, Paraguay, Peru, Poland, Qatar, Romania, Saudi Arabia, Serbia, Singapore, Slovenia, Spain, Sri Lanka, Sweden, Switzerland, United Arab Emirates, United Kingdom, United States, Venezuela, Zimbabwe, Afghanistan, Albania, Algeria, American Samoa, Angola, Anguilla, Antarctica, etc.